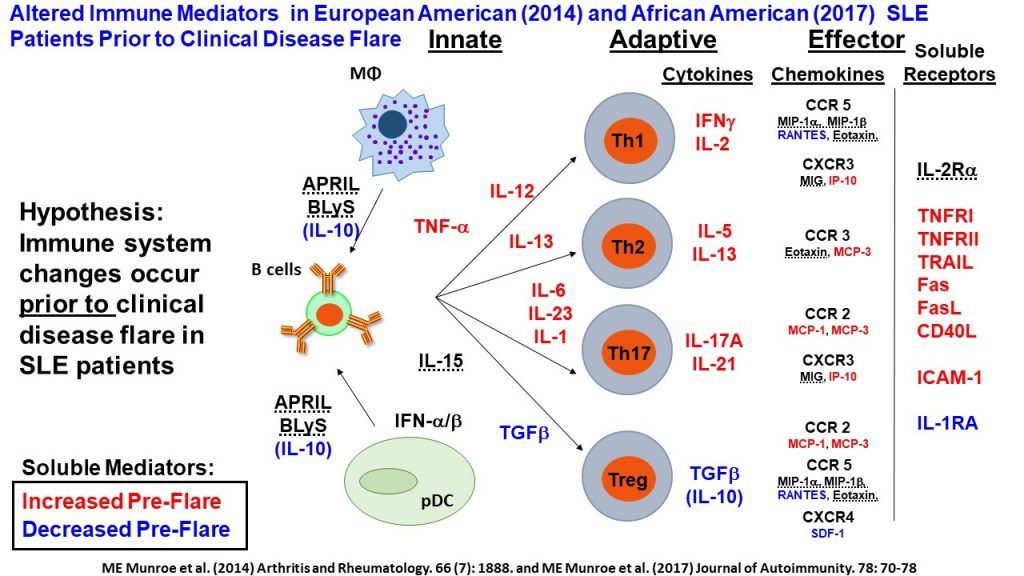
 Patients with lupus and other systemic autoimmune diseases often experience periods of relatively stable disease punctuated by periods of intense disease activity. These periods of intense disease activity, called flares, cause substantial morbidity and organ damage. To enable earlier detection and optimized treatment of flares, we study the immune disturbances that precede and lead up to clinical flares.
Patients with lupus and other systemic autoimmune diseases often experience periods of relatively stable disease punctuated by periods of intense disease activity. These periods of intense disease activity, called flares, cause substantial morbidity and organ damage. To enable earlier detection and optimized treatment of flares, we study the immune disturbances that precede and lead up to clinical flares.
We identify the immune changes associated with disease activity through a unique combination of clinical and mechanistic research. The Oklahoma Cohort for Rheumatic Diseases follows participants as they are seen in the research clinic for routine and emergent rheumatic disease care. This powerful resource has up to 16 years of longitudinal clinical data, autoantibody data, and samples from over 650 SLE patients, 450 RA patients, and 60 undifferentiated connective tissue disease patients. Through modular expression analysis, high-dimensional protein assays, and machine learning with SLE samples from this cohort, we identified molecular signatures that distinguish seven subsets of SLE patients and correspond with disease activity.
We are now validating these molecular signatures in independent cohorts through investigator-initiated studies in the research clinic and clinical trials in the Oklahoma Autoimmunity Center of Excellence. Further analyses are building on these findings to deconstruct the molecular heterogeneity and pathogenic mechanisms that are disease-specific and those that are shared across autoimmune diseases. These studies combine high-dimensional cytometry, single-cell transcriptomics and proteomics, transcriptomics, and high-throughput testing of soluble biomarkers. We then use translational informatics approaches to visualize these complex datasets, understand disease mechanisms, and identify potential biomarkers that may inform future clinical trials, treatment selection, and disease monitoring.
Browse other areas:
Pre-clinical Autoimmunity
Deciphering Molecular Heterogeneity
Improving Health Equity
Human Immunity to Infection and Vaccines